
Breast cancer remains one of the most prevalent malignancies and a leading cause of mortality among women worldwide. BRCA1 and BRCA2 are critical tumor suppressor genes responsible for repairing damaged DNA. Harmful variants in these genes significantly increase the risk of breast, ovarian, and other cancers such as fallopian tube, peritoneal, and prostate cancer. With NGS becoming a cornerstone in genetic testing, BRCA1/2 variant detection is accelerating bringing a parallel rise in Variants of Uncertain Significance (VUS) that demand precise classification.
Genetic testing for BRCA1/2 provides clarity on cancer predisposition, enabling early diagnosis and guiding personalized treatment and management strategies for improved outcomes. G2M BRCA1/2 NGS Assay ensures complete coverage and high confidence detection of critical BRCA1/2 mutations, ensuring proactive intervention and precision oncology. The panel is suitable for breast cancer detection and diagnosis identifying both germline and somatic mutations in the whole CDS (+/-40bp) and promoter regions of breast cancer associated BRCA 1 & BRCA 2 genes with high specificity.
*The panel’s performance is from the Illumina platform.
Proven Consistency in BRCA 1/2 Coverage
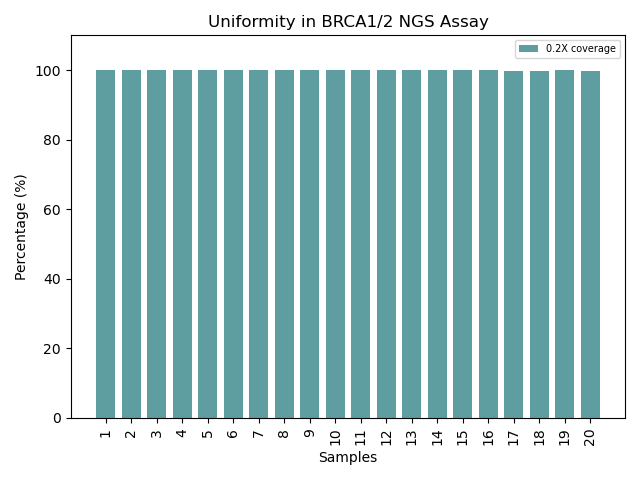
Exceptional Uniformity for BRCA1/2 Coverage: BRCA1/2 NGS assay demonstrates near-perfect uniformity, with 0.2X coverage consistently maintained across 20 samples, ensuring reliable variant detection and assay robustness.
Mutation Landscape: BRCA Genes in Patient Samples
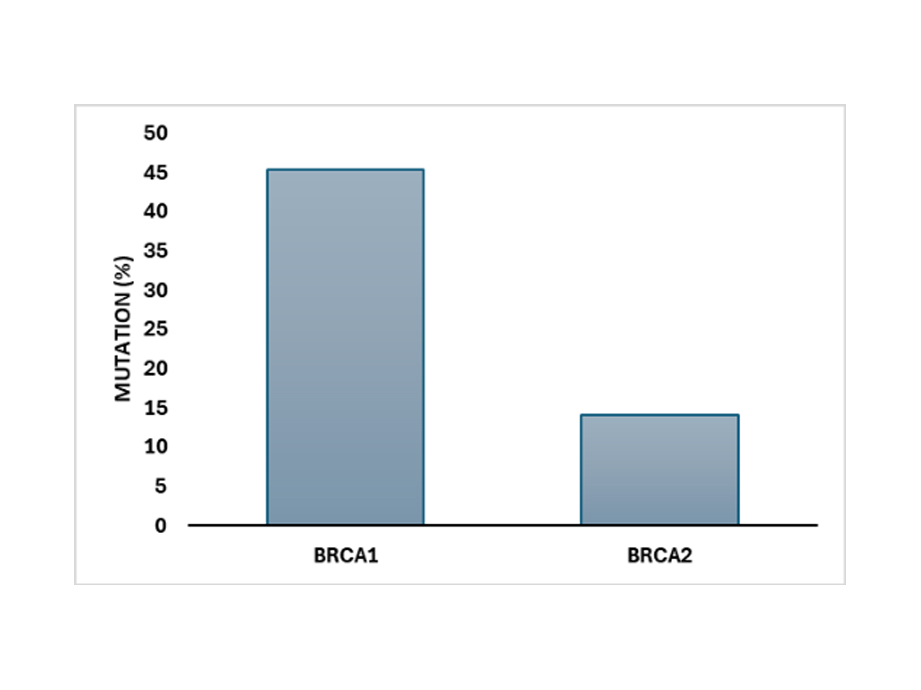
Comparative Mutation Frequency in BRCA Genes: Mutation frequency detected by the G2M BRCA 1/2 NGS Panel across patient samples shows BRCA1 has a significantly higher mutation prevalence (~45%) compared to BRCA2 (~14%), highlighting the critical role of BRCA mutations in hereditary breast and ovarian cancer risk assessment.
| Commercial Name | Old Cat No. | New Cat No. | Pack Size | Platform |
|---|---|---|---|---|
| BRCA NGS Test Kit | G2MBR00001 | G710001-1 | 24 T | Illumina |
| G2MBR00001 | G710001-2 | 96 T | Illumina | |
| G2MBR00001 | G710001-3 | 96 T – EZY | Illumina – EZY | |
| BRCA NGS Test Kit | G2MBR00001 | G710001-4 | 24 T | MGI |
| G2MBR00001 | G710001-5 | 96 T | MGI | |
| G2MBR00001 | G710001-6 | 96 T – EZY | MGI – EZY | |
| BRCA NGS Test Kit | G2MBR00001 | G710001-7 | 24 T | Aviti |
| G2MBR00001 | G710001-8 | 96 T | Aviti | |
| G2MBR00001 | G710001-9 | 96 T – EZY | Aviti – EZY | |
| BRCA NGS Test Kit | G2MBR00001 | G710001-10 | 24 T | Thermo |
| G2MBR00001 | G710001-11 | 96 T | Thermo | |
| G2MBR00001 | G710001-12 | 96 T – EZY | Thermo – EZY |
Since its inception in 2016, Genes2me has been constantly striving towards setting a benchmark in the diagnostics space by introducing premium quality (Made in India) diagnostic kits which are CE-IVD, ISO-13485:2016, and ISO 9001:2015 certified, assuring our clients of unparalleled quality and compliance with international standards.
© 2025 Genes2me. All rights reserved.