Closing the Diagnostic Gap in Tuberculosis: Why Molecular Diagnostics Matters More Than Ever

On World TB Day, conversations often focus on awareness. However, one of the most critical challenges in tuberculosis (TB) control is delayed and inadequate diagnosis. Despite being preventable and curable, TB continues to affect millions worldwide. A key reason? Many cases are either missed or diagnosed too late, allowing the disease to spread and worsen patient outcomes. Advanced molecular diagnostics, including real-time PCR, point-of-care systems, and targeted NGS are transforming TB diagnosis by enabling faster detection, comprehensive resistance profiling, and more effective, timely treatment decisions.
Globally, TB burden remains highest in South Asia, Southeast Asia, and Sub-Saharan Africa. India alone accounts for a significant share of cases. Many countries across Africa and Asia continue to face gaps in healthcare access, infrastructure, and diagnostic reach.
Despite decades of control programs and standardized treatment guidelines, TB continues to strain healthcare systems. The challenge today is not just access to diagnostics, but the growing complexity of the disease itself.
Conventional diagnostic methods such as smear microscopy, culture-based testing, and established molecular assays have long served as gold standards. These tools play a critical role in identifying active TB and guiding treatment decisions. Many national programs rely on them within structured diagnostic pathways.
However, these approaches have clear limitations. Smear microscopy lacks sensitivity in low bacterial load and extrapulmonary cases. Culture methods, although reliable, take weeks to deliver results. Even established molecular tests may not provide complete resistance information. These gaps delay treatment decisions and allow continued transmission.
At the same time, the TB landscape has evolved. The disease is no longer defined only by detection challenges. It is now shaped by increasing complexity, driven largely by the rise of drug-resistant strains.
The Growing Threat of Drug Resistance
TB is evolving, and drug resistance is at the centre of this challenge.
Drug-resistant TB (DR-TB), including multidrug-resistant (MDR), pre-extensively drug-resistant (Pre-XDR), and extensively drug-resistant (XDR) forms, has significantly complicated disease management. These forms require longer treatment, are harder to treat, and often lead to poorer outcomes.
In many cases, conventional diagnostics confirm TB infection but fail to identify resistance early. As a result, patients may receive ineffective therapy. This delays recovery and increases the risk of transmission within communities.
Moreover, resistance is no longer limited to first-line drugs such as rifampicin and isoniazid. Increasing resistance to second-line and newer drugs has added another layer of complexity.
This evolving landscape highlights a clear need- diagnostics must go beyond detection. They must enable early, accurate, and comprehensive resistance profiling.
Strengthening Diagnosis: The Role of Molecular Testing

Molecular diagnostics has emerged as a cornerstone of modern TB detection. These technologies enable rapid, sensitive, and highly specific identification of Mycobacterium tuberculosis. More importantly, they provide insights into genetic mutations linked to drug resistance.
Real-time PCR (RT-PCR) assays have significantly improved turnaround time. They allow faster diagnosis and support early clinical decision-making. These assays also perform well in low bacterial load cases, improving detection rates.
Genes2Me’s TB-Q Comprehensive Real Time PCR Kit enables simultaneous detection of Mycobacterium tuberculosis and key multidrug resistance mutations. It targets genes such as rpoB, katG, and inhA, enabling early identification of resistance to rifampicin and isoniazid.
The MTC-Q Real Time PCR Kit provides sensitive detection of Mycobacterium tuberculosis complex. Meanwhile, the MTB-NTM Multiplex Real Time PCR Kit differentiates TB from non-tuberculous mycobacterial (NTM) infections, an essential distinction for accurate clinical management.
Expanding Access: The Rise of Point-of-Care Testing

While accuracy is critical, access and speed are equally important, especially in high-burden and resource-limited settings.
Point-of-care testing (POCT) brings diagnostics closer to patients. It enables faster decision-making and improves access in decentralized settings.
Integrated platforms that combine nucleic acid extraction and RT-PCR offer streamlined, sample-to-result workflows with minimal manual intervention.
Genes2Me’s OnePCR automated detection system integrates extraction and amplification in a closed system. It reduces manual handling, minimizes contamination risk, and delivers rapid, reliable results with automated analysis.
However, the critical need today is to understand which drugs the infection is resistant to and how early this information can be obtained.
Moving Beyond Detection: The Power of Next-Generation Sequencing
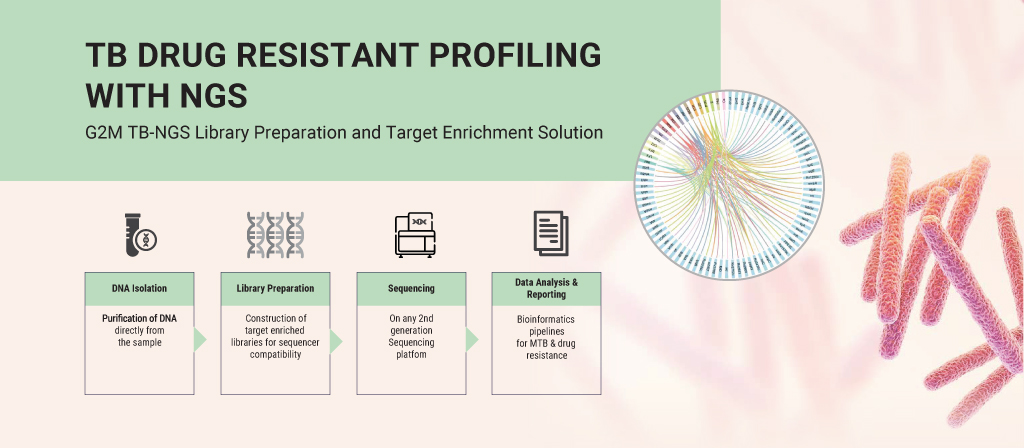
To address this challenge, advanced genomic technologies such as targeted next-generation sequencing (tNGS) are transforming TB diagnostics.
Targeted sequencing enables comprehensive analysis of genetic regions linked to drug resistance. It allows clinicians to make informed treatment decisions early in the diagnostic journey.
Genes2Me’s TB NGS Library Preparation and Target Enrichment Solution maps a 100 kb region of the Mycobacterium tuberculosis genome. It covers 75 drug-resistance genes and associated mutation sites, enabling detailed resistance profiling directly from clinical samples.
This hybridization capture-based approach eliminates the need for time-consuming culture methods. It improves turnaround time while providing deeper, more accurate insights.
By covering resistance across first-line, second-line, and newer WHO-recommended drugs, NGS supports personalized and effective treatment strategies.
Building a Comprehensive Diagnostic Ecosystem for TB Control
As the global fight against tuberculosis enters a new phase defined by drug resistance and complex clinical presentations, a single diagnostic modality is no longer sufficient.
From rapid detection using RT-PCR, to decentralized testing through POCT, and comprehensive resistance profiling with NGS, a multi-layered diagnostic approach is essential to effectively manage TB in today’s landscape.
Our portfolio reflects this integrated vision- combining speed, accessibility, and precision to support healthcare providers across different stages of TB diagnosis and management.
Looking Ahead
Ending TB will require more than treatment. It will require precision diagnostics that can keep pace with an evolving pathogen. As healthcare systems worldwide intensify efforts to eliminate TB, advanced molecular technologies will play a central role in detecting drug resistance, guiding therapy, and preventing further transmission. Strengthening access to these technologies, particularly in high-burden regions, will be key to turning global TB elimination goals into a reality.
At Genes2Me, we are committed to supporting this shift through innovative molecular diagnostic solutions designed to enhance accuracy, speed, and accessibility in TB detection.
On this World TB Day, we reaffirm our commitment to advancing diagnostics-because closing the diagnostic gap is key to ending TB.
