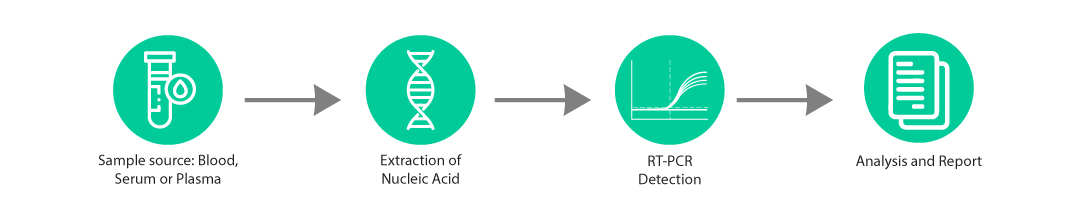
The Para-Q Multiplex kit is an in-vitro molecular diagnostic test, based on real-time PCR technology, for the detection of targets specific to Dengue, Malaria & Chikungunya along with Internal Control in clinical samples.
Malaria Specific Primer and Probe Mix | Chikungunya Specific Primer and Probe Mix | Dengue Specific primer and Probe | Internal Control Primer and Probe | One step 5x qRT-PCR Master Mix | Internal Control Template | RNase/DNase free water

Compatibility with all Common Platforms

Reiable Result with Endogenous Internal Control

Extensively Validated on wide variety of Clinical Specimens

Quick
Turnaround Time

High Sensitivity & Specificity

CE-IVD Certified Diagnostic Test
● Compatible with a wide range of 4 or 6 Fluorescence Channel Real-Time PCR devices and Genes2Me RapiCycler 96 Real Time PCR System
● Compatible with the Genes2Me’s RAPi-Q and RAPi-Q HT Point-of-Care (POC) RT-PCR Systems
CAT # |
DESCRIPTION |
UNITS |
G2M706121 |
Para-Q Multiplex Real Time PCR Kit |
100 Rxn |
G2M706121 |
Para-Q Multiplex Real Time PCR Kit |
50 Rxn |